Spain has taken an unprecedented step in the health field by applying gene therapy for individuals with a rare disease known as Butterfly Skin for the first time. Two patients from Andalusia have started using the Vyjuvek drug in public hospitals for this treatment, opening the doors to a new era in the treatment of dystrophic epidermolysis bullosa.
This development, long awaited by families and associations, has been made possible through an extraordinary mechanism for access to medications. This process will continue until the evaluation and funding at the state level are completed. While this news brings both relief and hope, it also presents a significant challenge: ensuring access to treatment for everyone with this disease, regardless of their autonomous region.
First Patients: Leo and Adrián
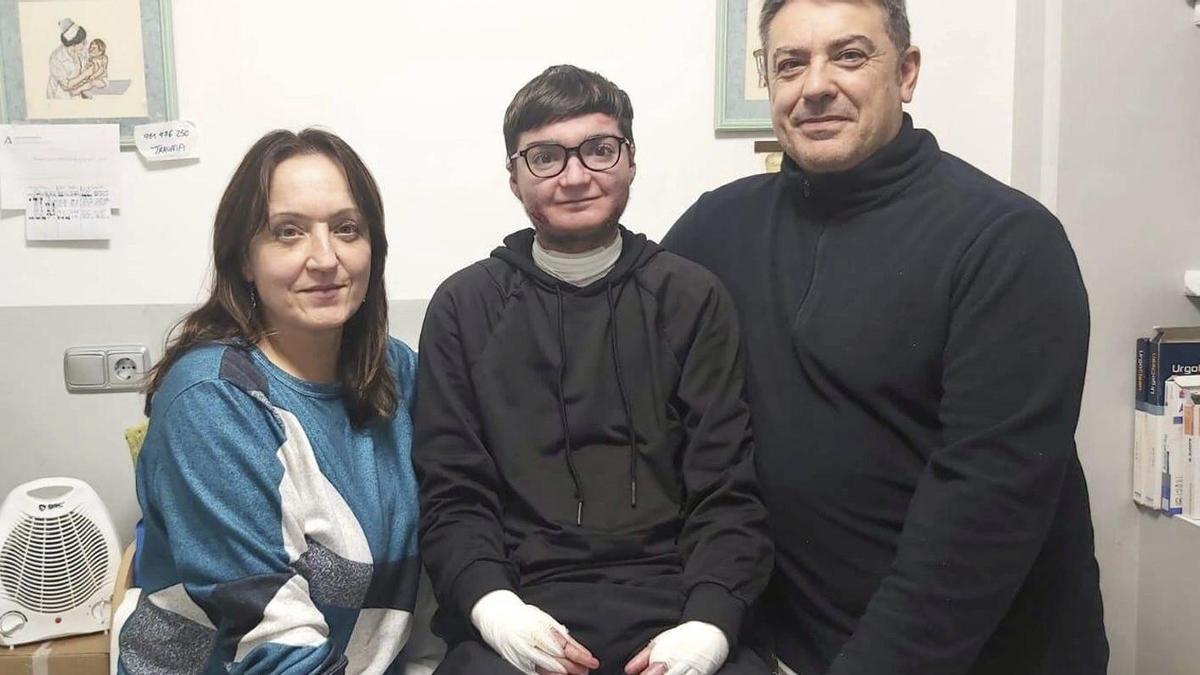
The first Spaniards to receive Vyjuvek are 12-year-old Leo from Seville and 22-year-old Adrián, a resident of Tolox (Málaga). Both have started treatment at the Virgen del Rocío hospital in Seville and the Costa del Sol hospital in Marbella, which are reference centers for this rare disease in Andalusia.
Leo has been living with dystrophic epidermolysis bullosa since birth. This condition requires him to deal with open wounds, blisters, and painful dressings every day. His story was brought to public attention through a video he made to accelerate the process for access to treatment, reaching as far as the European Parliament.
Adrián has been battling the consequences of this disease for over 20 years; it restricts his mobility, makes daily actions like dressing or walking difficult, and requires constant management of infections and wounds. Starting treatment means a long-awaited opportunity for him and those around him.
Leo's mother, Lidia, summarized the feelings of many families by saying “this treatment will change our lives” and expressed hope that the treatment would reach others affected in Spain as soon as possible. These words reflect years of waiting, bureaucracy, and the institutional pressures to make this drug a reality.
Before starting treatment, Leo stated he was “excited and eager”, realizing that “twelve years have passed waiting for this moment”. For a child who has to start a day filled with gas and bandages, the possibility of reducing pain and dressing changes means a significant change in his daily routine.
What is Butterfly Skin and Why is it So Restrictive?
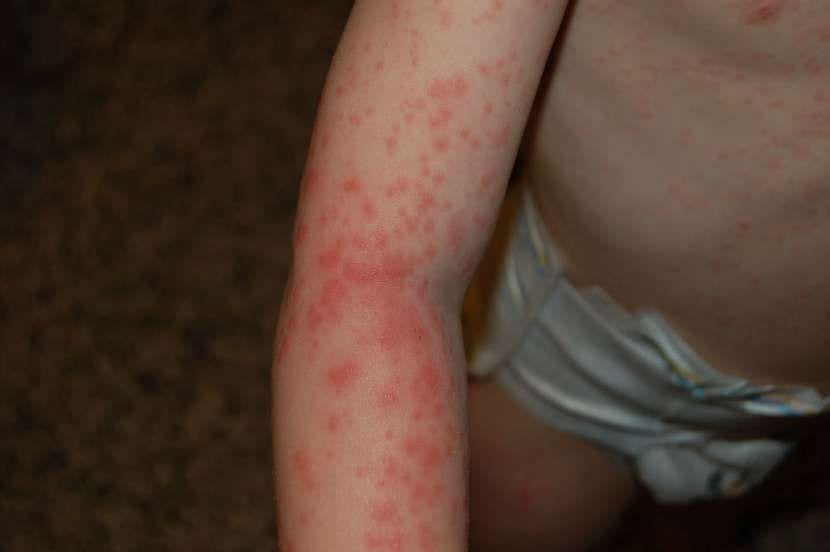
Epidermolysis bullosa, more commonly known as Butterfly Skin, is a rare genetic disease that creates extreme fragility in the skin and, in many cases, mucous membranes. A simple friction, a slightly tight shoe, or a piece of clothing that moves too much can cause blisters and wounds similar to third-degree burns.
In the dystrophic form experienced by Leo and Adrián, the problem lies in a gene that prevents the dermis and epidermis from “sticking together properly”. As a result, skin layers easily separate, painful blisters appear, and wounds can take a very long time to heal or may never close.
In daily life, this results in long and complex dressing changes lasting two to five hours a day. Leo's family explains that they need to wake him up at seven in the morning, clean and dress his body before he goes to school, and leave his legs and arms for the evening. Contact with water, changing bandages, or removing gas that has stuck to the skin can be particularly painful.
Beyond physical pain, the disease also has a strong emotional and social impact. Chronic wounds, scars, restrictions in joints, and fear of friction affect relationships with others and restrict autonomy and freedom of movement. Many children with this disease grow up with a constant sense of fragility and need help for almost every activity.
Until Vyjuvek and other research lines arrive, the approach to Butterfly Skin was primarily focused on symptomatic and supportive treatment: dressings, infection control, pain management, physical therapy, environmental adaptations, and psychological support. Therefore, offering a treatment targeting the genetic root of the problem signifies a significant shift in approach.
Vyjuvek: The First Gene Therapy for Epidermolysis Bullosa Dystrophica
Vyjuvek is the commercial name of the first gene therapy drug specifically developed for epidermolysis bullosa dystrophica. This is a topical treatment that involves a modified virus to introduce a functional copy of the defective gene (COL7A1) directly to skin wounds.
This gene is responsible for producing type VII collagen, a key protein for the adhesion of skin layers. By providing a functional version of the gene, the drug aims to promote the formation of new binding structures between the dermis and epidermis, which increases the skin's resilience and healing ability.
In practice, Vyjuvek is applied to wounds at specific intervals, under expert supervision, to accelerate the closure of chronic wounds, reduce the formation of new blisters, and alleviate pain associated with dressings. Although it is not a definitive treatment, existing data indicate significant improvements in the evolution of specific wounds and the overall quality of life of treated individuals.
In a disease where each dressing can last for hours and recurring infections and complications are common, any reduction in the number, size, and duration of wounds has a direct impact on autonomy, comfort, and the ability to perform daily activities such as going to school, working, or simply dressing with fewer restrictions.
The European Medicines Agency (EMA) approved this treatment, and subsequently, the European Commission issued an authorization allowing member states to begin incorporating it into their health systems. Spain has started to use it in a controlled manner until public funding is finalized.
Possibility of Use in Spain
The application of Vyjuvek in Spain was carried out through a special access mechanism for drugs; this is a pathway provided for innovative treatments that have not yet been included in the National Health System services portfolio. However, it applies to treatments with regulatory approval and sufficient evidence.
In this context, some autonomous regions chose to take steps ahead of others, undertaking the purchase of the drug and establishing specially trained teams. Andalusia has led as the first region to treat Butterfly Skin patients with gene therapy.
Health professionals from four Andalusian hospitals -Córdoba, Sevilla, Málaga, and Almería- have received special training to appropriately administer this drug, and after obtaining the necessary permissions, the first cases have been treated.
Meanwhile, the Ministry of Health is actively negotiating for the inclusion of Vyjuvek in the National Health System drug services. The laboratory developing the drug, Krystal Biotech, submitted an official funding request in March 2026, and the agreement to be reached will be a decisive factor in ending the dependency on extraordinary formulas for access.
Until this process is completed, the use of the treatment in Spain will be limited to case-by-case decisions and the willingness of autonomous regions to utilize existing extraordinary mechanisms. Therefore, it is currently applied only in those regions that activate these procedures.
DEBRA and the Key Role of Families
Behind the arrival of this treatment in Spain are the years of work by the DEBRA Butterfly Skin association and affected families. The process began in 2022 with the first contacts with the responsible company for the drug, and since then, institutional and social pressure has continued to grow.
In 2025, with the support of the European Medicines Agency and before the European Commission completed the process, DEBRA intensified its actions with hundreds of letters and meetings with the Ministry of Health, General Cortes, autonomous regions, and the Spanish Medicines and Health Products Agency. The goal was clear: to expedite the access of Vyjuvek to Spanish hospitals.
The citizens' movement has also been decisive. A campaign launched in October 2025 managed to surpass 150,000 support signatures, and these signatures were delivered to the Minister of Health. Additionally, in February 2026, two affected families, including Leo's, brought the demand to the European Parliament, drawing attention to a little-known but highly restrictive disease.
In this context, Leo's situation created a special impact. Videos explaining what it is like to live with open wounds and interventions in front of Spanish and European institutions helped the Butterfly Skin to transition from an invisible reality to becoming part of political and media discussions on therapeutic innovation.
DEBRA reminds that the arrival of Vyjuvek is not an end, but a stage in a journey that continues to support families, promote new research lines, and advocate for advanced treatments not to be limited to just a few hospitals or regions.
Demand for Unequal Access and Regional Equality
Although the treatment of Butterfly Skin with gene therapy in Spain is a significant milestone, the patient association emphasizes that access to treatment is still unequal among autonomous regions. In practice, its use depends on individual approvals in each region, creating very different situations based on residence.
Evanina Morcillo, the director of DEBRA Spain, emphasized that these first cases are “a promising step, but not enough” and reminded that receiving a life-changing treatment should not depend on a postal code. The organization demands equality in access to both Vyjuvek and future advanced treatments.
Andalucía has been among the leading regions by quickly activating the access mechanism in special cases, training its professionals, and taking on initial funding. Other regions, such as the Canary Islands, have started to mobilize, but still maintain uncertainty regarding access to the medication for families in other regions of Spain.
Until negotiations with the Ministry of Health are resolved and it is clarified whether the medication will be included in the National Health System's common portfolio, many Butterfly Skin patients are still on waiting lists and awaiting administrative decisions from the reference hospitals where they will receive treatment.
The association emphasizes that the inclusion of Vyjuvek in drug services should be carried out with clear and homogeneous protocols for the entire country, thus eliminating access disparities between regions and ensuring that the treatment is applied safely and coordinately in centers with the necessary experience.
Impact on Daily Life and Future Expectations
The impact of this treatment on families is measured with very concrete details: less dressing time, less pain when removing a bandage, a decrease in the fear of a simple hug. Leo's mother explains that he has had chronic wounds on his neck and armpits for ten years, making it impossible to feel direct air in those areas or wear certain t-shirts due to the fear of blood or serum stains.
Reducing the number of open wounds, shortening healing times, and decreasing infections can mean that a child goes to school more regularly, plays with other children with fewer restrictions, or simply sits on a chair without worrying about whether their clothes will stick to their skin. These are changes that do not always reflect in statistics but completely transform daily life.
Expectations regarding Vyjuvek are high, but also moderate. Those who have experienced this treatment in other countries explain that results can vary depending on the individual and the treated area. Some families have seen significant improvements within weeks, but in other cases, progress is slower. In any case, the general perception is that this is not a definitive treatment but a tool in the therapeutic arsenal.
In parallel, Butterfly Skin continues to be the subject of various research lines, such as other gene therapies, cellular treatments, or combination approaches. The medium- and long-term goal is not only to improve symptoms but also to intervene fundamentally in the genetic cause of the diseases.
Until this scenario comes to fruition, the launch of Vyjuvek in Andalusia's public health and the funding process in the rest of Spain represents a significant milestone. For many patients like Leo, Adrián, and those with Butterfly Skin, there is a feeling that a door that has been closed for years is beginning to open, and the efforts of associations, professionals, and families are starting to bear fruit.
The initiation of gene therapy for Butterfly Skin in Spain symbolizes a shift in the treatment of this rare disease: it combines significant scientific progress, sustained social movement, and increased administrative commitment. While there are still steps to be taken to ensure access and reinforce funding, the fact remains that the first patients now have a therapeutic option that can alleviate pain, reduce endless dressing times, and offer a more liberated life.


































Comments
(2 Comments)